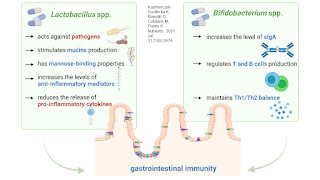
In the past few decades, Lactobacillus and Bifidobacterium genera were the main bacteria to be used as probiotics. One of the reasons for such attention was that these bacteria have the ability to thrive aerobically and are simple to package for sale. Cass Nelson-Dooley and Tony Hoffman of Metametrix (acquired by Genova Diagnostics in 2012) talked about these probiotics in MEBO interviews in 2009, emphasizing that high levels of Lactobacillus could be bad in some cases. Diets rich in simple carbs, poor absorption and small intestinal pockets can feed and trap excess Lactobacillus. Adding probiotics Bifidobacteria or Saccharomyces boulardii and prebiotic fiber to the diet could, in their opinion, help the MEBO population. D-lactate was one of the tests they offered that measured the level of this byproduct of bacterial fermentation, indicating an overgrowth of bacteria in the small intestine. According to our early clinical studies, D-lactate, however, was elevated in less than 20% of MEBO and PATM populations.
Even though Lactobacillus is only a minor member of the human colonic microbiota, the proportions of those bacteria and particular strains are frequently either positively or negatively correlated with human disease and chronic conditions. Although it generally is not considered a pathogen, it can cause disease in compromised hosts, including bacterial endocarditis, pleuropulmonary infections, gastrointestinal abscesses, urinary tract infection, conjunctivitis, dental caries, and endometritis. Lactobacillus bacteremia, presumably secondary to bacterial translocation from the gastrointestinal tract, has been reported in a patient with severe intestinal inflammation caused by ulcerative colitis. A decrease in genus Faecalibacterium and increase in Lactobacillaceae has been previously reported in patients with constipation (Lactiplantibacillus plantarum P9 supplementation, on the other hand, helped to increase frequency of bowel movements, while L11 improved gut health and odor in cats). Compared to healthy individuals, pediatric and adult Crohn's disease (CD) patients had an increased relative abundance of Lactobacillus species. In both cases, Lactobacillus enrichment coincided with depletion of F. prausnitzii - that could represent a good candidate as next-generation probiotic. Another study found that Weissella cibaria ZWC030 can inhibit skatole.
The percentages of Bifidobacterium and the Lactobacillus group presented a decreasing trend in patients with quiescent ulcerative colitis (UC) compared to active UC, although no significant differences were observed. Excessive amounts of Bifidobacteria can have negative effects on the body, such as in the case of Bifidobacterium breve bacteremia caused by excessive probiotic supplementation in infants with gastrointestinal conditions. On the other hand, Lactobacillus, Bifidobacterium and F. prausnitzii are depleted in IBS patients, resulting in lower SCFA concentrations (short-chain fatty acids, which are organic acids produced by gut bacteria during fermentation of indigestible foods) compared with healthy individuals. Maternal prenatal stress could also cause lower levels of Lactobacillus and Bifidobacterium and keep them low even later in life. With lower levels of Bifidobacterium, celiac patients have an imbalance in the intestinal microbiota, regardless of pH, even while on a gluten-free diet. Hormone treatment affects Bifidobacterium longum's ability to produce acids: progesterone reduces lactic acid and acetic acid production, while estradiol and thyroxine increase levels of both lactic acid and acetic acid (ethinyl estradiol) or lactic acid and butyric acid (thyroxine hormone).
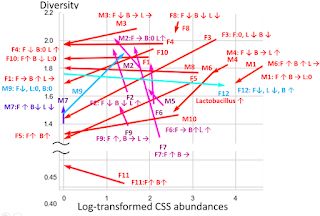
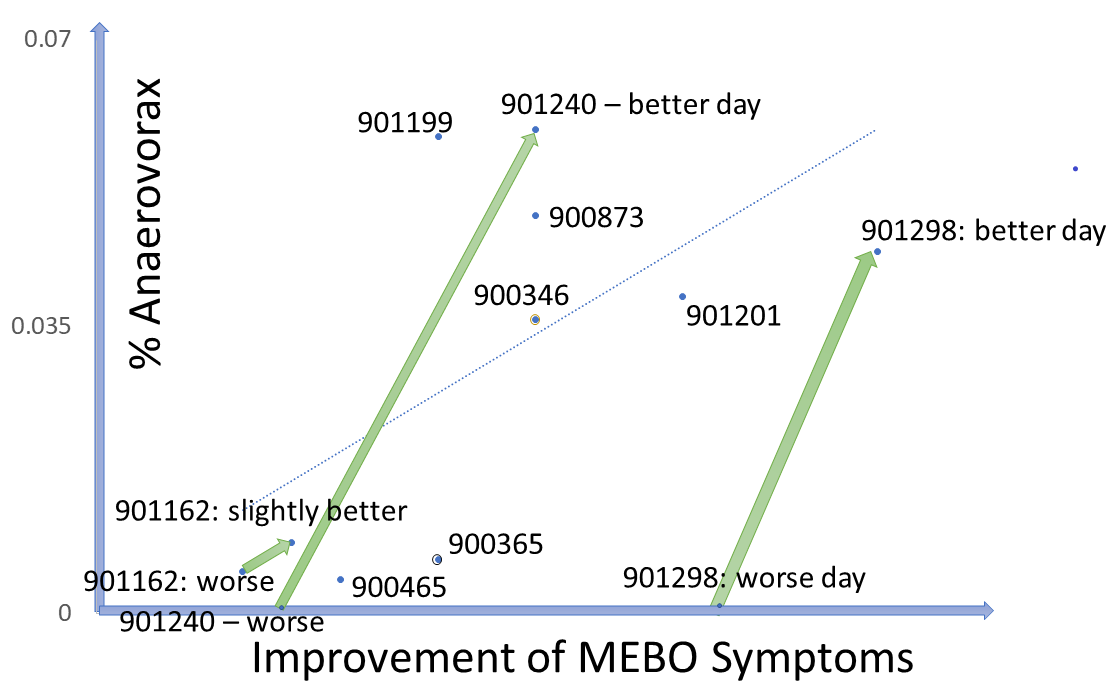
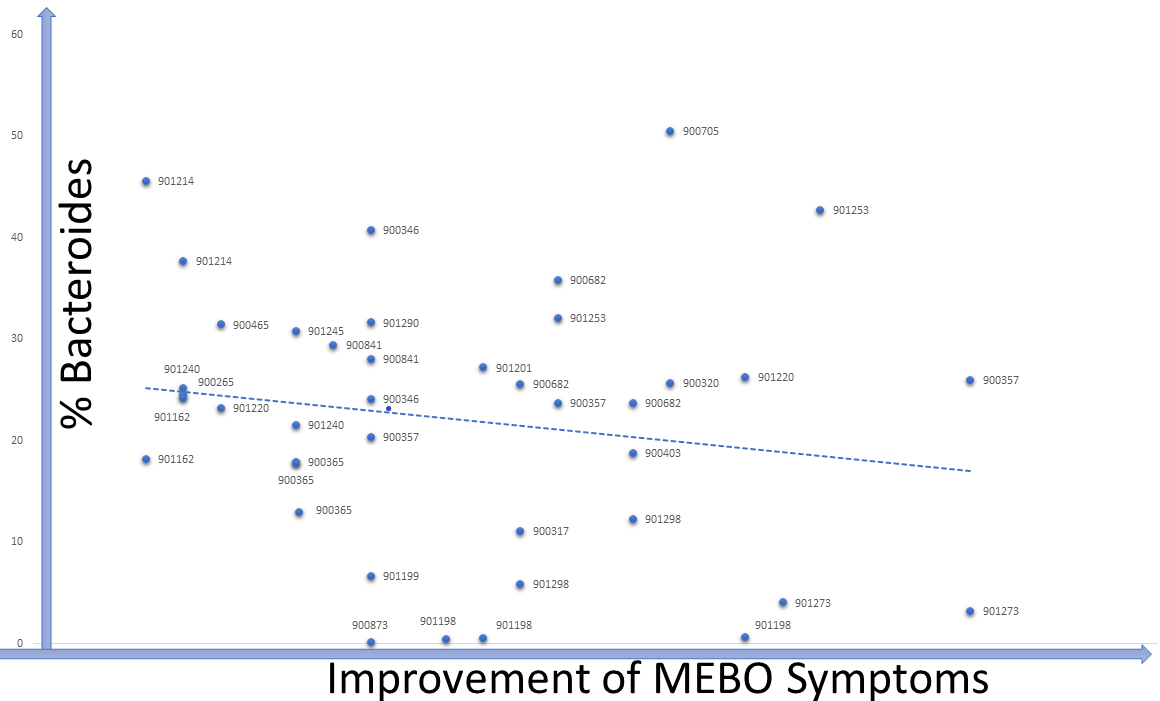
Data from our microbiome study (NCT03582826: Microbial Basis of Systemic Malodor and "People Allergic To Me" Conditions - ClinicalTrials.gov) show that while there was a general trend of increasing levels of Bifidobacteria and Lactobacillus in individuals who achieve remission, less than 10% of participants needed to boost levels of both species to improve their condition. In line with previous knowledge, in MEBO/PATM population, Bifidobacteria (one of the first microbes to colonize the human gastrointestinal tract) was more common in younger individuals. About one third of the participants experienced lesser symptoms when increasing levels of their gut Bifidobacteria, another third benefited from more Lactobacillus and the remaining third felt better when increasing levels of Faecalibacterium prausnitzii (a butyrate-producing anaerobe typically associated with good health). Changes in the levels of these bacteria corresponding to recovering are overlayed on Figure 5 from our paper published in JMIR dermatology. It was illustrating changes in microbial diversity vs abundances of selected bacterial species associated with odor (CSS) for 12 female (F1-F12) and 10 male (M1-M19) participants who self-reported both flare-ups (beginning of the arrow) and improvements (end of the arrow). We added text boxes showing whether the levels of F. prausnitzii (F), Bifidobacterium (B) and Lactobacillus (L) are increasing (🡹) , decreasing (🡻) or fluctuating (🡺) as symptoms resolve. It is obvious that individuals with the highest bacterial diversity don't benefit from increasing levels of probiotic diversity, while those with low diversity levels do need more probiotic microbes in their system. The figure demonstrates the complexity of these bacterial perturbations and emphasizes the need to consider more than just three species when predicting which treatment will work best for reducing MEBO and PATM symptoms.As we mentioned in the previous blog post, Lactobacillus is the main genera responsible for odors of the uncooked food waste and off-flavor pit mud. Some strains of Lactobacillus produce lactic acid, which can contribute to a more acidic environment in the gut. This can be beneficial for some people, but for others it can exacerbate odor issues. Bifidobacteria could also produce organic acids such as acetic acid and lactic acid in addition to short-chain fatty acids (SCFAs) like butyrate, which can have anti-inflammatory effects and promote gut health, but some people may find it unpleasantly smelling. F. prausnitzii produces a range of metabolites including one of the worst smelling compounds Putrescine. F. prausnitzii plays important role in balancing immunity. Its butyrate production has been correlated to the capacity to induce IL-10, an anti-inflammatory cytokine, in peripheral blood mononuclear cells (PBMC) but not to the ability to block IL-8 secretion in TNF-α-stimulated HT-29 cells. In COVID-19, Faecalibacterium prausnitzii has been inversely correlated with disease severity.
Research has shown that the gut microbiome is far more complex than previously thought, with hundreds of bacterial species interacting in a delicate balance. Therefore, focusing solely on Lactobacillus and Bifidobacterium, or any other bacterium, may not be enough to promote a healthy gut microbiome for everyone. Besides, different strains of bacteria within the same species can have different effects on the body.
After more than a century of research, the following species are identified as the primary probiotic species of Lactobacillus species: L.acidophilus, L.casei, L.fermentum, L.gasseri, L.johnsonii, L.paracasei, L. plantarum, L. rhamnosus, and L. salivarius. For Bifidobacteria these are: B.adolescentis, B.animalis, B.bifidum, B.breve, and B.longum. We are learning more about strains of these species. Novel probiotic strain Bifidobacterium bifidum CECT 7366 is active against the pathogenic bacterium Helicobacter pylori - and so are variations of Saccharomyces Boulardii and Lactobacilli johnsonii. Another strain L. plantarum 299v can directly interfere with E. coli colonization and improve the immunological status of the intestinal mucosa although these results have not yet been confirmed in humans. Lactobacillus GR-1 and B-54 or RC-14 strains in the vagina has been shown to reduce the risk of urinary tract infections. Mileti et al. found that Lactobacillus paracasei displayed a delay in the development of colitis and a decreased severity of disease but that L. plantarum and L. rhamnosus GG exacerbated the development of dextran sodium sulfate (DSS)-induced colitis.
L. rhamnosus supplementation contributes to higher intestinal absorption of free choline and elevated production of methylamines including TMA, whereas L. paracasei consumption may result in increasing bacterial consumption of choline for cholesterol assimilation and phospholipid metabolism rather than for methylamine metabolism.
Bifidobacterium supplemented with various fiber additives can prevent the growth of Clostridium difficile, while Lactobacillus salivarius protects the broiler chicks from Salmonella infections. Several strains of other species promising as probiotics are from Roseburia spp., Akkermansia spp., and Faecalibacterium spp. Probiotic potential risk score (PPRS) allowed to classify 84 Faecalibacterium prausnitzii strains into low-, medium-, and high-risk groups. 15 strains identified as low-risk strains are prioritized for clinical application.
Unfortunately, the extreme oxygen sensitivity of F. prausnitzii imposes practical challenges to the production, transportation, storage, and manufacturing of probiotic products to be evaluated in a clinical setting. Instead, we have to turn to prebiotics - a type of dietary fiber - in our food . F. prausnitzii's growth is promoted by inulin, inositol (vitamin B8) and fructo-oligosaccharides, chicory roots, wheat, onion, banana, garlic, and leek, wheat, rye, rice, barley, oat, and sorghum, and gold kiwifruit.
Gut microbiota can be also influenced by phytochemicals. In one study, black raspberry (BR) reduced the serum levels of trimethylamine-N-oxide and cholesterol in rats fed excessive choline with a high-fat diet (HFC). The authors hypothesized that since gut microbiota plays a crucial role in the production of trimethylamine and microbial metabolites, BR could influence gut microbial composition. Their study examined microbiomes and metabolomes of rats and showed that the BR supplementation enriched Bifidobacterium and reduced Clostridium cluster XIVa.
While we have made significant progress in understanding the role of probiotics and prebiotics in supporting our gut health, it's important to recognize that there is still a great deal that we don't know. It's becoming increasingly clear that a personalized approach to prebiotic and probiotic use may be necessary to achieve optimal health outcomes. This approach involves paying attention to how different foods make you feel and adjusting your diet accordingly, while eating a diverse and balanced diet that incorporates a variety of whole foods and phytochemicals, staying hydrated, getting enough sleep and exercise, and managing stress levels. Focusing on overall health and well-being will help to support the body's natural ability to maintain a healthy microbiome and avoid dysbiosis-related health issues.
REFERENCES
Din AU, Hassan A, Zhu Y, Yin T, Gregersen H, Wang G. Amelioration of TMAO through probiotics and its potential role in atherosclerosis. Applied Microbiology and Biotechnology. 2019 Dec;103:9217-28.
Pacifico L, Osborn JF, Bonci E, Romaggioli S, Baldini R, Chiesa C. Probiotics for the treatment of Helicobacter pylori infection in children. World J Gastroenterol 2014; 20(3): 673-683 [PMID: 24574741 DOI: 10.3748/wjg.v20.i3.673]
Lim T, Lee K, Kim RH, Ryu J, Cha KH, Park SY, Koo SY, Hwang KT. Effects of black raspberry extract on gut microbiota, microbial metabolites, and expressions of the genes involved in cholesterol and bile acid metabolisms in rats fed excessive choline with a high-fat diet. Food Sci Biotechnol. 2023 Feb 13;32(4):577-587. doi: 10.1007/s10068-023-01267-4. PMID: 36911337; PMCID: PMC9992478.
Heeney DD, Gareau MG, Marco ML. Intestinal Lactobacillus in health and disease, a driver or just along for the ride?. Current opinion in biotechnology. 2018 Feb 1;49:140-7.