Cytochrome P450, often abbreviated as CYP450 (CYP) or simply P450, is a vital group of enzymes found in the liver, and it plays a fundamental role in detoxifying the body and metabolizing various foreign compounds.
Metabolic enzymes employ different catalytic mechanisms. FMOs, for instance, directly receive electrons from nictinamide adenine dinucleotide phosphate (NADPH), while CYPs obtain their electrons via an intermediary protein known as CYP reductase. Furthermore, CYPs activate oxygen only after binding to an oxygenatable substrate, adding an extra layer of complexity to the metabolic puzzle.
In the complex world of enzymatic metabolism, our comprehension of the comparative efficiency of different enzymes remains somewhat limited. The body's selection of which metabolic enzymes to use is governed by several factors such as substrate specificity, enzyme efficiency, temperature and the surrounding environment in the compartment where the enzyme and substrate are in, co-factors and co-enzymes, concentration of substrates and competition for substrates.
Cytochrome P-450 (CYP450) enzymes and Flavin-containing monooxygenases (FMOs), such as FMO3, often participate in the metabolic processes of the same compounds. For instance, substances like Nicotine, Caffeine, Tazarotenic acid, Benzydamine, and the antipsychotic drug Perazine (PER) encounter these enzymes during their metabolic journey.
FMO3, in particular, stands out for its remarkable ability to convert trimethylamine (TMA) into trimethylamine N-oxide (TMAO). However, even TMA navigates a maze of metabolic pathways and could encounter cytochromes P-450.
Similar to FMO3, genetic variations in CYP2E1, such as the SNP g.50657948 T>G, have been linked to odor (lamb odor and flavor in sheep), indicating a broader role beyond metabolism, potentially affecting odorant and pheromone clearance. Ubiquitous amino acid derived from food - tryptophan serves as the precursor for skatole, and the conversion of tryptophan to skatole involves the action of enzymes, including CYP2E1, in a series of metabolic reactions. CYP2E1 expression levels have been correlated with a variety of dietary and physiological factors, such as ethanol consumption, diabetes, fasting, and obesity.
Poor dietary choices, medications, exposure to external factors such as air pollution, cigarette smoke, radiation (e.g., UV radiation from the sun), and certain environmental toxins, inflammatory processes, whether due to infection, injury, or chronic inflammatory conditions, even normal metabolic processes can generate reactive oxygen species (ROS) as byproducts. Examples are superoxide anion (O2·-), hydrogen peroxide (H2O2), hydroxyl radical (·OH), and singlet oxygen (1O2), among others. Excessive production or impaired elimination of ROS can lead to oxidative stress.
To counteract the harmful effects of oxidative stress, the body activates the detoxification process, in which cytochrome P450 takes center stage. Cytochrome P450 catalyzes the addition of an oxygen atom to foreign compounds, making them more water-soluble. This transformation results in the formation of alcohols and, as byproducts, aldehydes. The detoxification process is a critical defense mechanism that helps the body eliminate harmful substances.
High-fat and high-protein diets have gained popularity but can have adverse effects on our health. Research has shown that these diets may induce organ damage, abnormal serum biochemical indexes, and inflammation. Interestingly, the production of malodorous gas compounds in the body because of these diets can be influenced by the interaction between the intestinal microbiota and liver cytochrome P450.
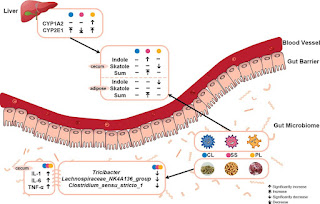 |
| from Zhang et al, 2022 |
Chemicals that alter xenobiotic metabolizing enzymes, such as CYPs, may also alter endogenous hormone levels since some of these enzymes control levels of endogenous hormones. Many of the pesticides that caused mammary gland tumors or other mammary effects also alter steroidogenesis in the H295R adrenocortical carcinoma cell line, activate nuclear receptors or CYP enzymes, or are estrogenic.
CYP family of heme monooxygenase enzymes is known for its ability to catalyze enantioselective hydroxylation and epoxidation reactions. Epoxidation reactions have been hypothesized to proceed via multiple mechanisms involving different reactive intermediates. A study of the bacterial enzyme CYP199A4 from Rhodopseudomonas palustris demonstrated a significant reduction in epoxidation activity when the D251N mutation was introduced. Remarkably, despite these mutations, the chemoselectivity and stereoselectivity of the epoxidation reaction remained intact.
Innovations in biotechnology have led to the development of specialized cytochrome P450 enzymes, such as the Cytochrome P450 BM-3 mutant (139-3). This mutant exhibits high activity towards the epoxidation of non-natural substrates, including propylene, which can be converted to propylene oxide.
In summary, cytochrome P450 is a fascinating and essential component of our body's biochemistry, with implications that extend beyond detoxification. Understanding its functions and interactions can pave the way for advancements in both medicine and biotechnology.
REFERENCES
Zhang T, Xie B, Liu H. High-fat and high-protein diets from different sources induce different intestinal malodorous gases and inflammation. Food Research International. 2022 Apr 1;154:110989.
Padwa A, Murphree SS. Epoxides and aziridines-a mini review. Arkivoc. 2006 Jan 1;3(6).
Störmer E, Brockmöller J, Roots I, Schmider J. Cytochrome P-450 enzymes and FMO3 contribute to the disposition of the antipsychotic drug perazine in vitro. Psychopharmacology. 2000 Sep;151:312-20.
Harahap RS, Noor RR, Gunawan A. Effect of CYP2E1 gene polymorphisms on lamb odor and flavor in Indonesian sheep. InIOP Conference Series: Earth and Environmental Science 2021 Jun 1 (Vol. 788, No. 1, p. 012022). IOP Publishing.
